For decades, biology textbooks have offered a simple description of mitochondria: they are the "powerhouse of the cell." While accurate, this moniker vastly undersells their influence. Emerging science reveals that these microscopic organelles are actually the arbiters of life and death, functioning as sophisticated environmental sensors that dictate our metabolic destiny.
We now understand that mitochondrial health is not just about having enough energy to get through the day. It is about the quality of that energy and the "exhaust" produced in the process. When mitochondria malfunction, they do not just stop working; they become active drivers of systemic inflammation, insulin resistance, and accelerated aging.
The Biological Hydroelectric Dam
To understand how to optimize vitality, we must first understand the machinery. Mitochondria generate Adenosine Triphosphate (ATP)—the currency of life—through a mechanism remarkably similar to a hydroelectric dam. They strip electrons from the food we eat and the oxygen we breathe, passing them through a series of protein complexes known as the electron transport chain.
This process pumps protons across a membrane to charge a microscopic battery. When these protons flow back through a turbine-like structure, they generate ATP. This relentless chemical industry powers everything from your heartbeat to your thoughts. However, like any industrial plant, this process generates waste.
The "Leaky" Mitochondria Theory of Aging
In a youthful, healthy system, the waste products of energy production—specifically reactive oxygen species (ROS)—are managed efficiently. But as we age, or under the burden of a sedentary lifestyle, mitochondria become "leaky."
A leaky mitochondrion is like an old car engine: it burns fuel inefficiently and spews out toxic black smoke. It produces less ATP while generating excessive free radicals. This state of chronic oxidative stress triggers inflammation and damages the cell from the inside out. Research identifies this dysfunction as a potential root cause for major metabolic conditions, including Type 2 diabetes and Alzheimer’s disease.[1]
By optimizing mitochondrial function, we are essentially tuning the engine to burn cleaner and last longer.
The Twin Pillars of Exercise: Density and Quality
Can we reverse this decay? The answer lies in hormesis—the biological principle that controlled stress strengthens the system. Exercise is the most potent hormetic stressor available, but not all movement affects mitochondria in the same way. Current research suggests a two-pronged approach.
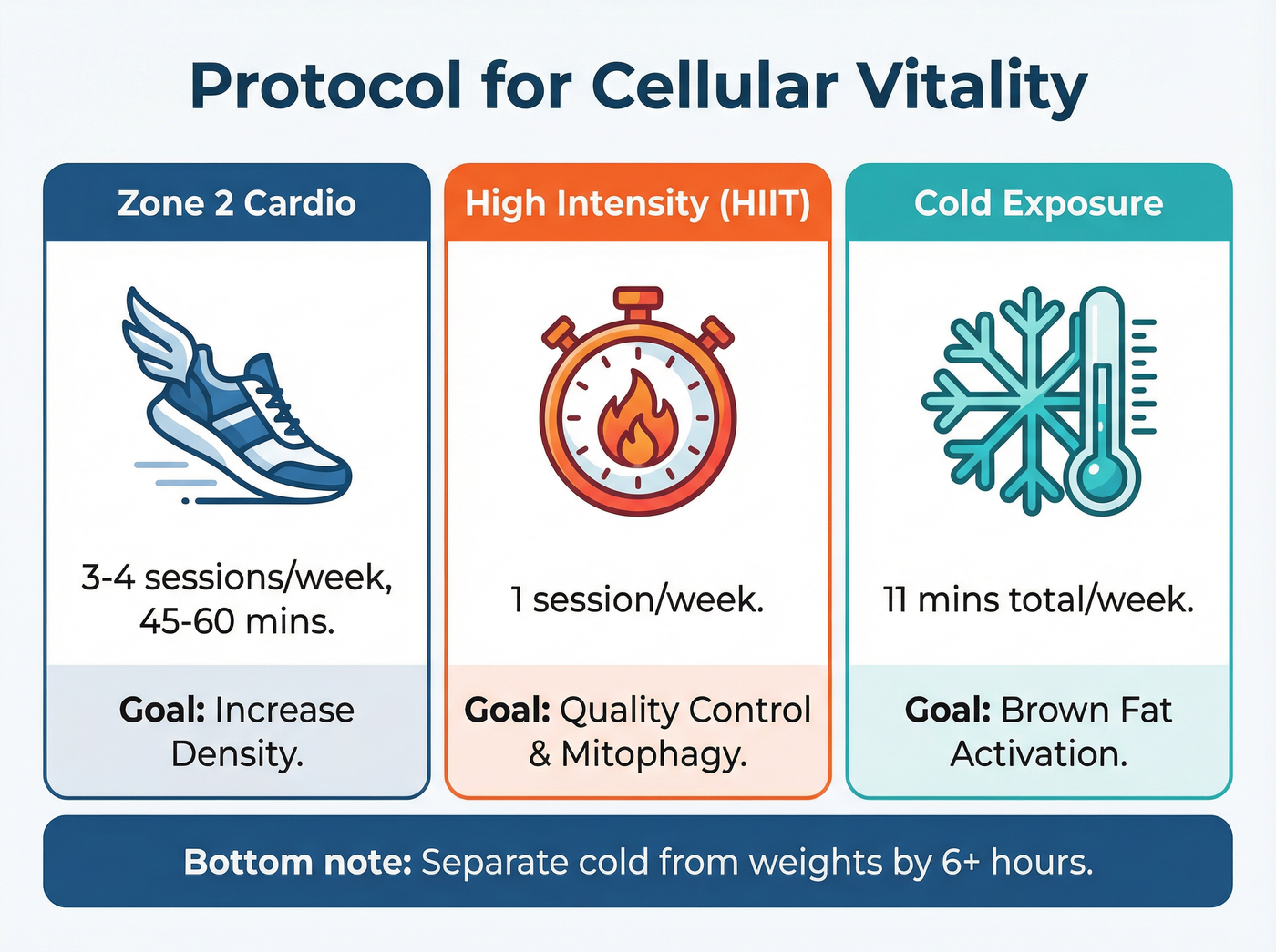
1. Zone 2 Training for "Base"
Low-intensity, steady-state cardio (often called "Zone 2") is where you can hold a conversation involved but are working hard enough to sweat. This type of training specifically targets mitochondrial density and efficiency in fat oxidation. It builds the infrastructure, increasing the sheer number of mitochondria in your slow-twitch muscle fibers.
2. High-Intensity Intervals for "Quality Control"
While low intensity builds the base, high intensity cleans the house. Recent systematic reviews highlight that vigorous exercise, such as Sprint Interval Training (SIT) or HIIT, creates a massive metabolic demand that triggers mitophagy—the recycling of old, broken mitochondria.[2][3]
Think of high-intensity efforts as a quality control mechanism. By forcing the cell into a temporary energy crisis, you signal the body to purge inefficient organelles and stimulate mitochondrial biogenesis (the creation of new ones). All-out sprints, even as short as 30 seconds, have been shown to rapidly boost VO2 max and mitochondrial content.[4][5]
Thermal Stress: The Cold Plunge Prescription
Beyond the gym, environmental stress is a powerful tool. Cold exposure—whether through ice baths or cold showers—forces the body to engage in non-shivering thermogenesis. This process is powered by mitochondria, specifically in brown adipose tissue (brown fat).
To leverage this for longevity, consistency is key. A popular protocol based on recent findings suggests an "11-minute rule": accumulating 11 minutes of cold exposure per week, divided into 2–3 sessions. The water should be uncomfortable (typically 50°F–59°F / 10°C–15°C), forcing the body to work to stay warm. This thermal shock boosts PGC-1alpha, a master regulator of mitochondrial growth.[6]
Critical timing note: If your goal is muscle growth, avoid cold immersion immediately after strength training, as it may blunt the inflammatory signals required for hypertrophy. Spacing them by 6–12 hours is the safer bet.
Designing a Protocol for Longevity
We are not passive recipients of our genetic age. Our mitochondria are highly plastic, meaning they change based on the signals we send them. Living a life of comfort sends a signal of decay; living a life that includes calculated discomfort sends a signal of survival and adaptation.
To build a resilient engine for the decades ahead, consider this weekly framework:
- Aerobic Base: 3 hours of Zone 2 activity (brisk walking, cycling, rucking).
- Peak Output: 1 dedicated session of high-intensity intervals (e.g., 4 x 4 minutes hard, or short all-out sprints).
- Thermal Stress: A cumulative 11 minutes of cold water immersion.
By treating your mitochondria like the environmental sensors they are, you improve not just your lifespan, but your healthspan—ensuring you have the energy to live fully, right up until the end.
Listen to the episode
Mitochondrial Health: The Science of Cellular Vitality
Sources
- How to Boost Mitochondrial Density and Quality for Longevity
- Healthspan Research Review | Volume or Intensity? A Systematic Review
- Effects of high-intensity interval training vs moderate-intensity continuous training on mitochondrial dynamics (2025)
- Effects of Exercise Training on Mitochondrial and Capillary Growth: A Systematic Review (2024)
- Exercise as Mitochondrial Medicine: Annual Review of Physiology (2025)
- The Key Role of Mitochondrial Function in Health and Disease