In the vast, blue expanse of the Mediterranean Sea, there exists a creature no larger than a pinky nail that has managed to achieve what human civilizations have dreamt of for millennia: eternal youth. While most organisms follow a strict linear path—birth, growth, reproduction, and death—a tiny hydrozoan has discovered a biological "backdoor." This is the story of Turritopsis dohrnii, the animal that can effectively hit the rewind button on its life cycle.
As of Saturday, January 24, 2026, scientists continue to study this unique creature to understand the fundamental mechanics of aging. By challenging the central dogma that cellular identity is fixed, the immortal jellyfish offers a glimpse into a world where biological time is not a one-way street. Its existence forces us to ask: Is aging an inevitable law of physics, or simply a software program that can be reset?
The Jellyfish That Cheats Death
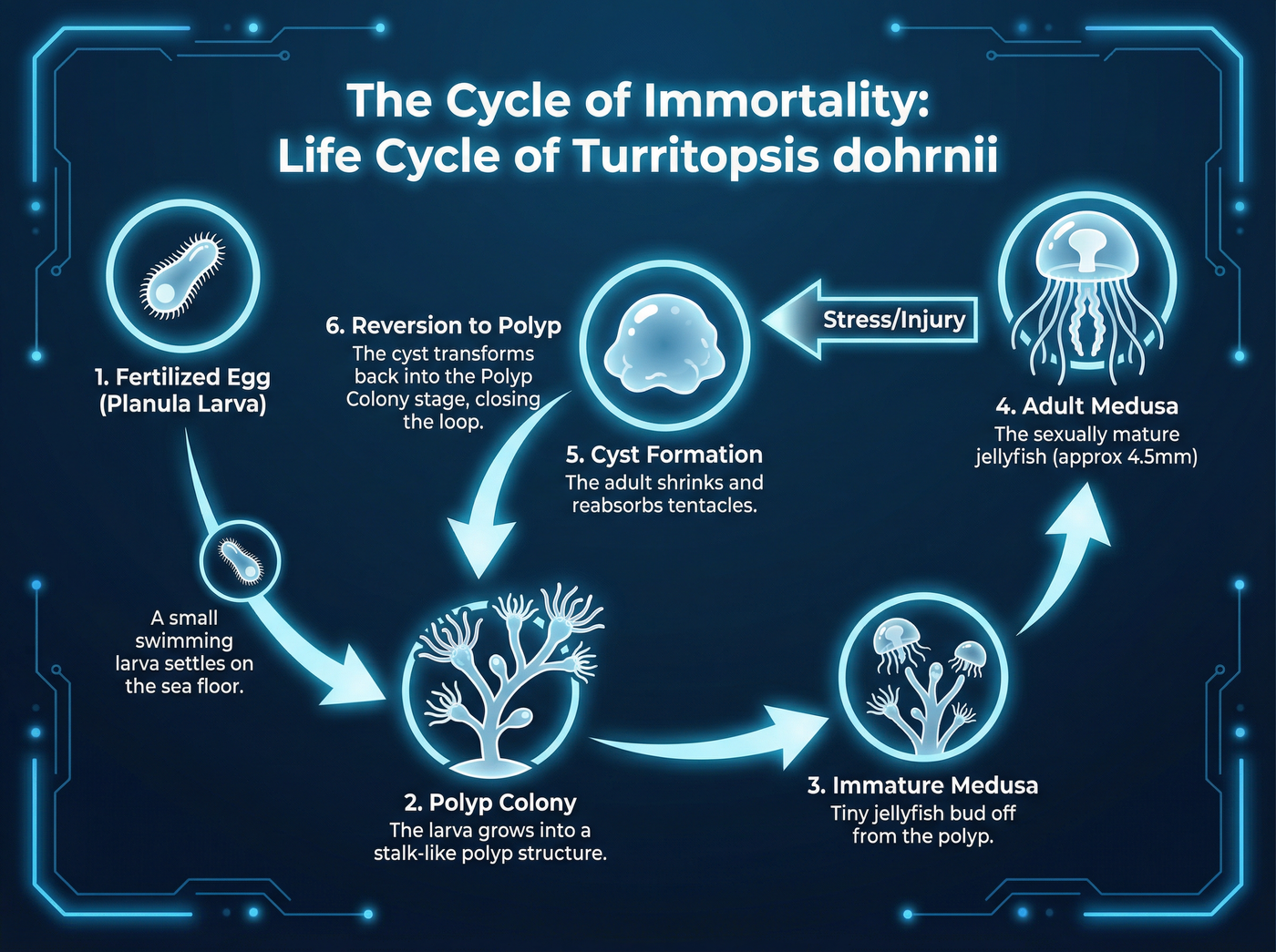
For centuries, biology operated under the assumption that development moves in a single direction. A fertilized egg becomes a larva, which becomes a polyp, and eventually a sexually mature medusa (jellyfish). Once an organism reaches maturity, the clock ticks toward senescence and death. The discovery of Turritopsis dohrnii shattered this rule.
When this jellyfish faces physical damage, starvation, or environmental stress, it refuses to die. Instead, it triggers a process of ontogeny reversal using a mechanism known as transdifferentiation.
Understanding Transdifferentiation
Transdifferentiation is the biological equivalent of turning a brick back into a lump of clay, and then reshaping that clay into a piece of wood. In most animals, once a cell specializes—becoming a muscle cell or a skin cell—its fate is sealed. However, the immortal jellyfish can take a specialized adult cell and transform it into a completely different cell type required for a younger life stage.[1]
During this transformation, the adult jellyfish retracts its tentacles, shrinks its bell, and sinks to the ocean floor, becoming a cyst-like blob. Within this cyst, the cells undergo a radical identity change, resetting the organism's biological clock. The cyst then develops into a polyp—the juvenile stage of the jellyfish—which eventually spawns a new colony of genetically identical clones.[4] This isn't just repair; it is biological reincarnation.
The process involves complex molecular mechanisms, including:
- DNA repair: Enhanced activity in genes responsible for fixing genetic damage.
- Telomere maintenance: High levels of telomerase activity prevent the shortening of protective chromosome caps, a primary driver of aging in humans.[2]
- Gene silencing: Turning off "adult" genes and reactivating "pluripotency" genes similar to those found in embryos.
The Undying Hydra
While the immortal jellyfish is famous for its dramatic reversals, another creature, the Hydra, takes a different approach to longevity. These tiny freshwater cnidarians do not appear to age at all. Instead of reversing their life cycle, they maintain a state of perpetual youth through constant cellular renewal.
The body of a hydra is composed almost entirely of stem cells. It replaces every cell in its body roughly every 20 days. Because of this, it does not accumulate cellular damage or senescence—the gradual deterioration of function characteristic of aging.[6]
Biologist Daniel Martínez conducted research demonstrating that the mortality rate of hydra remains constant over time. A hydra that has lived for years is no more likely to die the next day than one born a week ago. This defies the standard biological model where death risk increases exponentially with age.[6]

Implications for Human Aging
If nature has solved the problem of aging in these simple organisms, can humans replicate it? The short answer is: it's complicated. The same mechanisms that allow the jellyfish and hydra to regenerate—unlimited cell division and plasticity—are dangerously similar to the mechanisms of cancer in humans.
However, studying these creatures has identified specific genetic pathways that could lead to medical breakthroughs. For instance, recent genomic studies on Turritopsis dohrnii have highlighted specific genes involved in DNA replication and repair that are far more active than in related, mortal species.[1] Understanding how these animals silence "adult" programming without causing uncontrolled tumor growth could revolutionize regenerative medicine.
Why aren't we immortal?
Evolutionarily speaking, immortality is expensive. Most animals in the wild die from predation or disease long before old age sets in. Therefore, nature tends to favor organisms that invest energy in rapid reproduction rather than indefinite maintenance. The immortal jellyfish and hydra are exceptions because their regenerative abilities serve as a specific survival strategy against catastrophic environmental stress or injury.
While we cannot yet turn back our biological clocks to infancy, the study of these organisms shifts our perspective. Aging is not an unchangeable fate, but a biological process managed by specific cellular machinery—machinery that, in theory, can be tuned.
Listen to the episode
Listen to "Endless Youth: The Secrets of Biological Immortality" on Pody.fm
Sources
- Regenerative characteristics of the immortal jellyfish, Turritopsis dohrnii, and their potential implications for human aging
- Regeneration and Immortality: Aging of the Jellyfish Turritopsis dohrnii
- An “Immortal” Jellyfish Offers Clues into Biological Aging
- Are any animals truly ‘immortal’? These creatures defy biological time.
